CAS PubMed Article Google Scholar. A molecule is a group of two or more atoms held together by chemical bonds.

Carbon Dioxide Molecule Co2 Lewis Dot Cross Electronic Diagram Covalent Bonds Ball Stick Space Filling 3d Models Boiling Point Melting Point Doc Brown S Chemistry Revision Notes
Rare Earth single-atom catalysts for nitrogen and carbon dioxide reduction.

. The carbon atoms are numbered from the end closest to the OH group. A water molecule is released. The vapor pressure of.
Depending on context the term may or may not include ions which satisfy this criterion. Single-wall carbon nanotubes SWCNTs Single-wall carbon nanotubes are one of the allotropes of carbon intermediate between fullerene cages and flat graphene with diameters in the range of a nanometerAlthough not made this way single-wall carbon nanotubes can be. Dehydration synthesis reactions _____ whereas hydrolysis reactions _____.
The name is 68-dimethyl-3-decanol not 35-dimethyl-8-decanol. The molecule is decomposed into carbon dioxide and water. In quantum physics organic chemistry and biochemistry the distinction from ions is dropped and molecule is often used when referring to polyatomic ions.
Calculate the volume of CO₂ gas in L collected over water at 250 C when 251 g of calcium carbonate is added to excess hydrochloric acid if the total pressure is 911 mm Hg. That fixes the two methyl CH 3 groups at the sixth and eighth positions. In the kinetic theory of gases the term molecule is.
A water molecule is used. ACS Nano 14 10931101 2020. When solid calcium carbonate is reacted with aqueous hydrochloric acid the products of the reaction include aqueous calcium chloride liquid water and gaseous carbon dioxide.
A carbon nanotube CNT is a tube made of carbon with diameters typically measured in nanometers. Five carbon atoms in the LCC make the compound a derivative of pentane. When a sucrose molecule is decomposed to yield a glucose molecule and a fructose molecule A.
Two OH groups on the first and fifth carbon atoms make the.
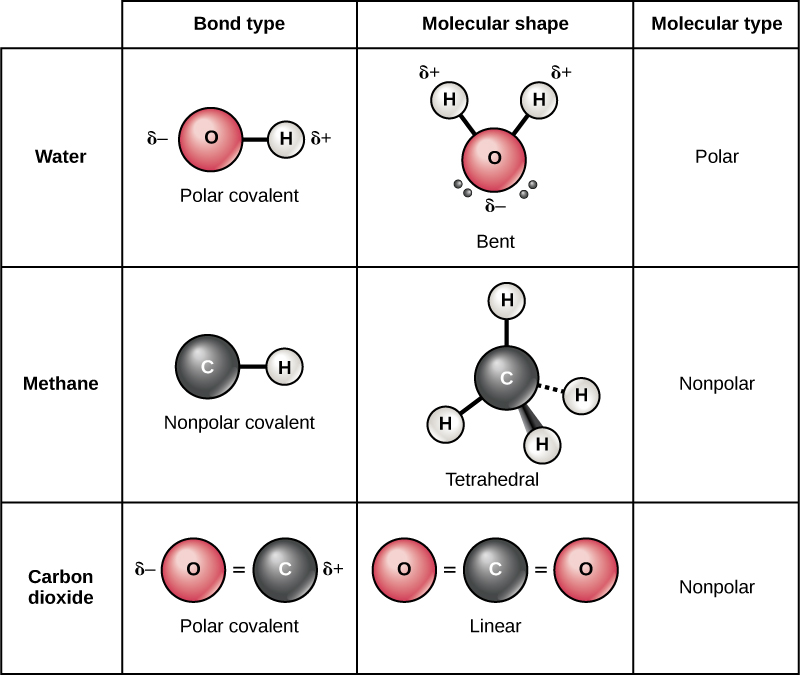
Covalent Bonds Biology For Non Majors I
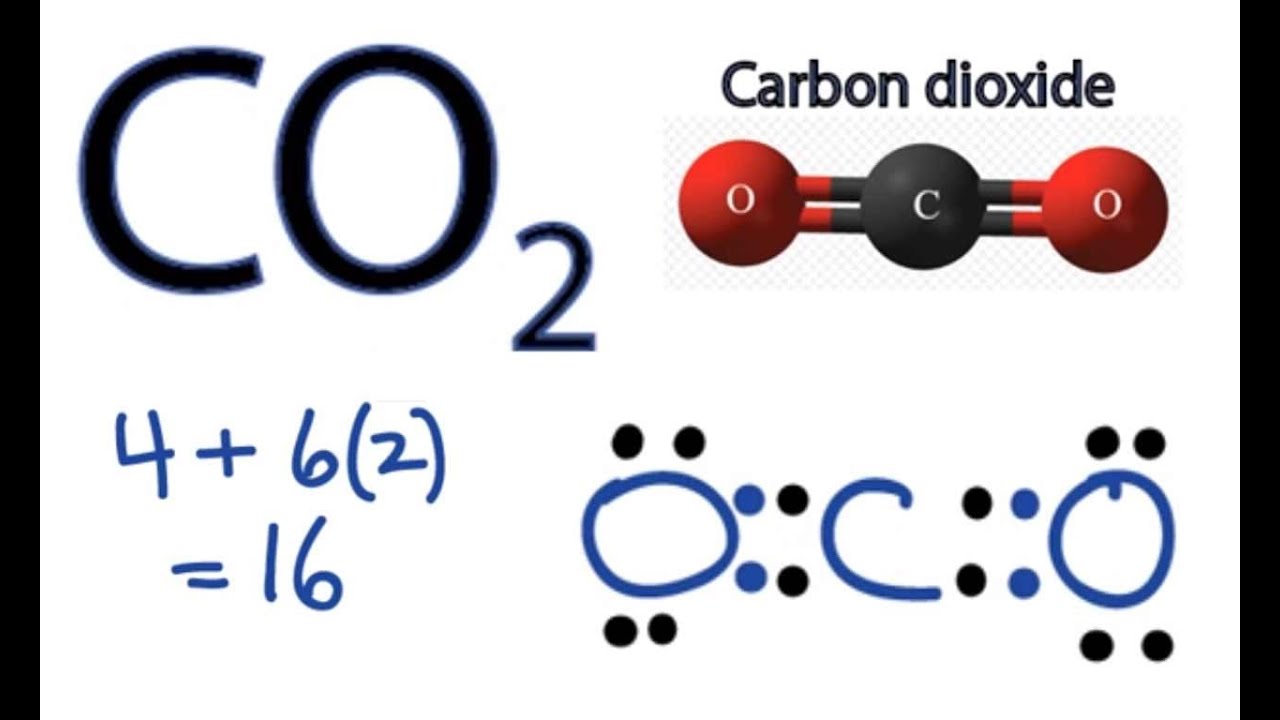
Co2 Lewis Structure How To Draw The Dot Structure For Carbon Dioxide Youtube

The Most Common Type Of Bond Formed By Carbon Is A Covalent Bond In Most Cases Carbon Shares Electrons W Chemistry Education Atomic Bonding College Chemistry
0 Comments